Abstract
PURPOSE
Urgency to initiate therapy has to be weighed against the time required for screening and consenting to enroll a patient onto a clinical trial. Exclusion of patients with rapidly progressive or symptomatic disease may lead to selection of patients with less aggressive disease. We recently identified that a short diagnosis to treatment interval (DTI) is associated with adverse clinical factors and inferior outcome in patients with diffuse large B-cell lymphoma (DLBCL) (Maurer et al, JCO 2018); shorter time to treatment was also found to be associated with outcome in other aggressive lymphomas, including mantle cell lymphoma (MCL), in the National Cancer Database (Olszewski et al, BJH 2018). Here we assess the association of DTI with clinical variables and outcome in MCL from an observational cohort with validation in patients enrolled on an NCTN clinical trial.
PATIENTS AND METHODS
Newly diagnosed patients with MCL were prospectively enrolled in the University of Iowa/Mayo Clinic Lymphoma SPORE Molecular Epidemiology Resource (MER), now a subcohort of the Lymphoma Epidemiology of Outcomes (LEO) Cohort Study. Patients initially managed with observation were excluded. DTI was defined as the time in days from date of first lymphoma-containing biopsy to the initiation of therapy. Associations of DTI with clinical factors and outcome were examined using a 14 day split based on the previous MER study of DTI in DLBCL. Odds ratios (OR) and Cox model hazard ratios (HR) are reported. Outcome was assessed using event-free survival (EFS) where events were defined as progression/relapse, initiation of new anti-lymphoma therapy, or death due to any cause. A validation cohort was assembled from patients enrolled in CALGB (Alliance) clinical trial 50403. Protocol therapy was high dose chemotherapy with autologous hematopoietic stem cell transplantation (ASCT) consolidation and post-ASCT rituximab followed by bortezomib as either consolidation or maintenance. DTI was defined as time from diagnosis to study registration and outcome was assessed using progression-free survival (PFS) as defined on the clinical trial.
RESULTS
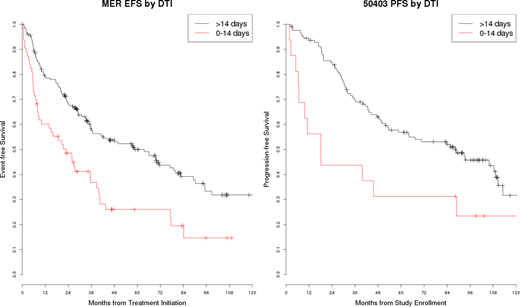
271 patients with MCL enrolled in the MER from 2002-2015 and initiated treatment within 100 days of diagnosis. Median age at diagnosis was 64 years (range 36-76), 211 (78%) were male, and 92% were stage III-IV; MIPI was Low (41%), Intermediate (36%) and High (23%). Induction therapy was R-CHOP based (50%), B-R based (16%), HyperCVAD based (8%), 2-CDA based (11%), and other (15%). Median EFS of the cohort was 41 months (95% CI: 34-46). Median DTI was 26 days (IQR: 14-38) and 28% enrolled within 14 days of diagnosis, exhibiting a longer DTI than DLBCL patients enrolled in the MER during the same time period. Short DTI (0-14 days) was associated with adverse clinical factors including ECOG PS 2-4 (OR=5.74, 95% CI: 2.51-13.1, p<0.0001), elevated LDH (OR=2.30 95% CI: 1.25-4.22, p=0.0072), and higher MIPI score (OR=1.48, 95% CI: 1.04-2.07, p=0.025) but was not significantly associated with age, sex, B-symptoms, or stage (all p>0.30). Short DTI was also associated with inferior EFS (HR=1.78, 95% CI: 1.27-2.49, p=0.0008) which remained significant after adjusting for MIPI score (HR=1.72, 95% CI: 1.23-2.41, p=0.0016).
In the validation cohort, 144 patients enrolled on 50403 within 100 days of diagnosis from 2006-2010. Median age at diagnosis was 59 years (range 29-69), 77% were male, and 97% were stage III-IV; MIPI was Low (51%), Intermediate (31%) and High (17%). Median PFS of the cohort was 84 months (95% CI: 50-109). Median DTI was 35 days (IQR: 22-46) and 11% enrolled within 14 days of diagnosis. Short DTI (0-14 days) was associated with PS (OR=9.00, 95% CI: 1.18-69.0, p=0.034) and) higher MIPI score (OR=2.09, 95% CI: 1.08-4.04, p=0.029) but not elevated LDH (OR=1.27, 95% CI: 0.43-3.74, p=0.66). Short DTI was associated with inferior PFS (HR=2.21, 95% CI:1.19-4.09, p=0.011) which remained clinically significant after adjusting for MIPI score (HR=1.85, 95% CI: 0.98-3.47, p=0.056).
CONCLUSION
A short diagnosis-to-treatment interval is strongly associated with adverse clinical factors and poor outcome in newly diagnosed MCL in the MER. Clinical trials are encouraged to facilitate enrollment of patients with short DTI to avoid potential selection bias.
Support: U01 CA195568, P50 CA097274, U10CA180821, U10CA180882; ClinicalTrials.gov Identifier: NCT00310037 (CALGB 50403)
Maurer:Celgene: Research Funding; Nanostring: Research Funding; Morphosys: Research Funding. Kaplan:Bayer Pharmaceuticals: Consultancy. Cohen:Celgene: Consultancy, Membership on an entity's Board of Directors or advisory committees; Janssen: Research Funding; Pharmacyclics: Consultancy, Membership on an entity's Board of Directors or advisory committees; Infinity Pharmaceuticals: Consultancy, Membership on an entity's Board of Directors or advisory committees; Bristol-Myers Squibb: Research Funding; AbbVie: Consultancy, Membership on an entity's Board of Directors or advisory committees; Seattle Genetics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Millennium: Consultancy, Membership on an entity's Board of Directors or advisory committees; Novartis: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; BioInvent: Consultancy; Takeda: Research Funding. Martin:Gilead: Consultancy; Kite: Consultancy; Seattle Genetics: Consultancy; AstraZeneca: Consultancy; Janssen: Consultancy; Bayer: Consultancy. Kahl:Abbvie: Consultancy; AstraZeneca: Consultancy; Gilead: Consultancy; Acerta: Consultancy; Juno: Consultancy; Celgene: Consultancy; Genentech: Consultancy; Seattle Genetics: Consultancy; CTI: Consultancy; ADC Therapeutics: Consultancy. Bartlett:ImaginAB: Research Funding; Bristol-Meyers Squibb: Research Funding; Novartis: Research Funding; Immune Design: Research Funding; Janssen: Research Funding; Merck & Co: Research Funding; Forty Seven: Research Funding; Novartis: Research Funding; Pharmacyclics: Research Funding; Affimed: Research Funding; Astra Zeneca: Research Funding; Pharmacyclics: Research Funding; Acerta: Membership on an entity's Board of Directors or advisory committees; Celgene: Research Funding; Millennium: Research Funding; Genentech: Research Funding; KITE: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Gilead: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Seattle Genetics: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding; Pfizer: Consultancy, Membership on an entity's Board of Directors or advisory committees, Research Funding. Cerhan:Celgene: Research Funding; Jannsen: Other: Scientific Advisory Board; Nanostring: Research Funding. Leonard:Karyopharm: Consultancy; Biotest: Consultancy; Bayer: Consultancy; Pfizer: Consultancy; ADC Therapeutics: Consultancy; Novartis: Consultancy; AstraZeneca: Consultancy; Juno: Consultancy; BMS: Consultancy; Sutro: Consultancy; MEI Pharma: Consultancy; Genentech/Roche: Consultancy; Celgene: Consultancy; Gilead: Consultancy; United Therapeutics: Consultancy.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal